Structure And Synthesis Of Glucagon
Glucagon is a 29-amino acid peptide hormone predominantly secreted from the alpha cells of the pancreas. It is derived from the precursor proglucagon which can be processed into a number of related peptide hormones . Proglucagon is expressed in pancreatic islet alpha cells, intestinal enteroendocrine L cells, and to a minor extent in neurons in the brain stem and hypothalamus . Processing of proglucagon is undertaken by the processing enzymes prohormone convertase 1/3 and prohormone convertase 2 , respectively. In the pancreas, PC2 processes proglucagon to glucagon while processing of proglucagon in the intestine and the brain is undertaken by PC1 leading to the formation of glucagon-like peptide 1 and glucagon-like peptide 2 .
Effects Of Glucose And Adenine Nucleotides On Exocytosis Evoked By Trains Of Depolarizations
We next investigated the effects of glucose in response to a train consisting of 10 500-msec depolarizations from 70 to 0 mV in the absence of glucose and 6 min after the addition of 20 mm glucose . In the absence of glucose, the capacitance increase elicited by the train amounted to 54 ± 15 fF . After addition of glucose, the amplitude of the capacitance increase was stimulated 3.3-fold and averaged 178 ± 13 fF . Secretory granules in rat -cells, like other hormone-releasing cells, can be functionally subdivided into a reserve pool and a limited readily releasable pool , which undergoes rapid exocytosis upon stimulation . The exhaustion of the exocytotic response during the train is likely to reflect depletion of the RRP rather than inactivation of the Ca2+ current with resulting suppression of Ca2+-induced exocytosis. This notion is supported by the observation that the integrated Ca2+ current measured at the end of the train, when secretion had ceased, was reduced by only 24 ± 9% with respect to the first depolarization.
Triggers For Hormone Release :
When and why are hormones released ? Hormones are produced and released into the bloodstream by endocrine glands and certain other tissues in the body. Depending on the specific hormone and the physiological conditions, the immediate stimulus for the release of hormones is one of : Endocrine glands release hormones in response to one of the following stimuli: Specific molecules in the blood, e.g. certain minerals or nutrients . Stimulation by other hormones Stimulation by signals from the nervous system Note: The numbers 1-3 do not indicate sequence or importance, merely that these 3 factors stimulating production and release of hormones are discussed in this section . The control of hormone secretions in the form of their immediate secretion being stimulated by these 3 mechanisms is described in more detail below. 1. Specific molecules in the blood The presence or level of specific molecules in the blood, e.g. certain minerals or nutrients – sometimes referred to by more technical terms that have specific meanings in biochemistry such as electrolyte and metabolite. Low levels of calcium ions in the blood stimulate the parathyroid glands to release parathyroid hormone . Increasing levels of blood glucose contribute to stimulation of tContinue reading > >
You May Like: What Is Congress Mortgage Stimulus Program
What Blood Glucose Levels Can Indicate
Our bodies desire blood glucose to be maintained between 70 mg/dl and 110 mg/dl . Below 70 is termed . Above 110 can be normal if you have eaten in the past 2 to 3 hours. That is why your doctor wants to measure your blood glucose while you are fasting, when the level should be between 70 and 110.
Even after you have eaten, however, your glucose should be below 180. Above 180 is termed . If your blood sugar measures above 200 after drinking sugar water , then you are diagnosed with .
Four Ways to Take Insulin: https://www.cdc.gov/diabetes/basics/type-1-4-ways-to-take-insulin.html
American Diabetes Association Professional Practice Committee, Draznin B, Aroda VR, Bakris G, Benson G, Brown FM, Freeman R, Green J, Huang E, Isaacs D, Kahan S, Leon J, Lyons SK, Peters AL, Prahalad P, Reusch JEB, Young-Hyman D. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022 Jan 1 45:S125-S143. doi: 10.2337/dc22-S009. PMID: 34964831.
Glucagon and Other Emergency Glucose Products: https://diabetes.org/healthy-living/medication-treatments/glucagon-other-emergency-glucose-products
Glucagon: https://www.ncbi.nlm.nih.gov/books/NBK559195/
Zeigerer A, Sekar R, Kleinert M, et al. Glucagons Metabolic Action in Health and Disease. Comprehensive Physiology. 2021 11:1759-1783. doi:10.1002/cphy.c200013
Effects Of Glucose On Glucagon Release From Intact Islets Exposed To Diazoxide And Elevated K+
Effects of glucose on glucagon and insulin secretion from clamped intact rat islets. Batches of 10 intact rat islets were incubated for 30 min in KRBH buffer containing 30 mm K+, 250 m diazoxide , or normal KRBH buffer with 4.7 mm K+ and no diazoxide . Under both experimental conditions, glucagon and insulin release was determined in the absence and presence of increasing glucose concentrations . C, As in A, except that glucagon release was determined in 0 or 16.8 mm glucose in the absence and presence of 10 mm mannoheptulose . Values are mean ± se for 10 batches of islets. *, P< 0.05 **, P< 0.01.
You May Like: How Much Was The Second Stimulus Check
Effects Of K+ On Glucagon Release
The concept that, as for -cells, glucose stimulates ATP formation leading to KATP-channel closure, membrane depolarization, Ca2+ influx, and glucagon release is supported by the observation that increasing the external K+ concentration resulted in a progressive stimulation of glucagon secretion starting at 11 mm K+ . This contrasts to our earlier observation in mouse -cells where increasing the extracellular K+ concentration up to 15 mm resulted in inhibition of glucagon release and only K+ concentrations beyond 25 mm produced stimulation of secretion .
Effects of -conotoxin and nifedipine on high K+-induced glucagon release in single rat -cells. A, Glucagon release was measured in batches of FACS-isolated -cells during 30-min incubations at increasing extracellular K+ concentrations in the absence of glucose. B, Histogram summarizing effects of the N-type Ca2+-channel blocker -conotoxin and the L-type Ca2+-channel blocker nifedipine on glucagon release stimulated by increasing extracellular K+ concentrations from 5.6 mm to either 15 mm or 50 mm K+. Data are mean values ± se of five different experiments. *, P< 0.05 **, P< 0.01.
How Is Glucagon Controlled
Glucagon works along with the hormone insulin to control blood sugar levels and keep them within set levels. Glucagon is released to stop blood sugar levels dropping too low , while insulin is released to stop blood sugar levels rising too high .
The release of glucagon is stimulated by low blood glucose, protein-rich meals and adrenaline . The release of glucagon is prevented by raised blood glucose and carbohydrate in meals, detected by cells in the pancreas.
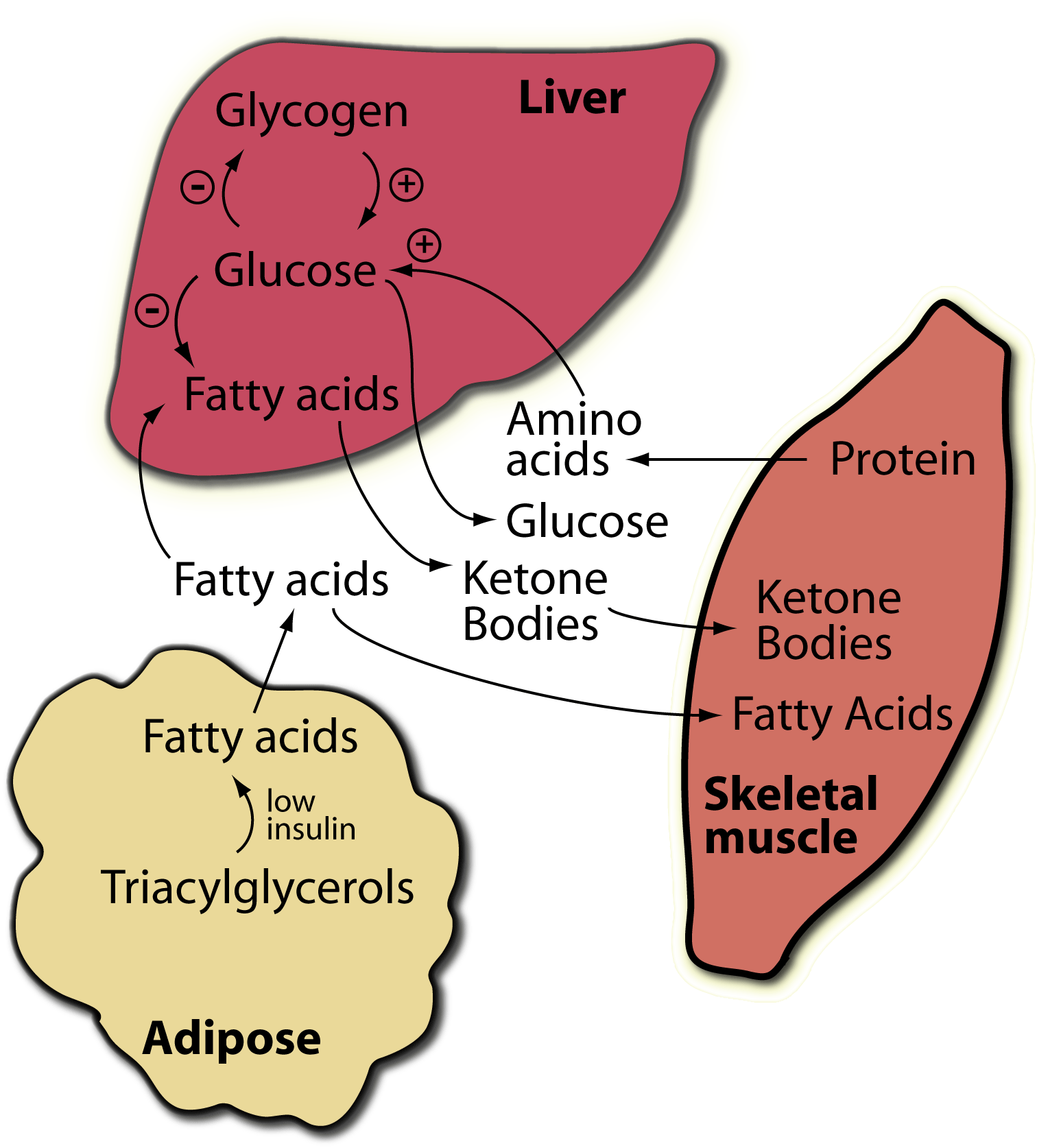
In the longer-term, glucagon is crucial to the bodys response to lack of food. For example, it encourages the use of stored fat for energy in order to preserve the limited supply of glucose.
Also Check: I Didn’t Get My California Stimulus Check
Glucagon Increases Hepatic Glucose Production
Glucagon controls plasma glucose concentrations during fasting, exercise and hypoglycemia by increasing hepatic glucose output to the circulation. Specifically, glucagon promotes hepatic conversion of glycogen to glucose , stimulates de novo glucose synthesis , and inhibits glucose breakdown and glycogen formation . Hepatic glucose production is rapidly enhanced in response to a physiological rise in glucagon achieved through stimulation of glycogenolysis with minor acute changes in gluconeogenesis . This ability of glucagon is critical in the life-saving counterregulatory response to severe hypoglycemia. Additionally, it is a key factor in providing adequate circulating glucose for brain function and for working muscle during exercise . During prolonged fasting, glycogen stores are depleted, and gluconeogenesis takes over . The hyperglycemic property of glucagon is enhanced when hepatic glycogen levels are high and diminished when hepatic glycogen levels are low in conditions of fasting or liver diseases like cirrhosis .
Intracellular Signaling Pathways Regulating Glp
Until recently, there has been a relative paucity of published literature on the intracellular pathways mediating the effects of GLP-1 secretagogues in the L-cell. However, the development of in vitro models of the murine, rat, and human intestinal L-cell has now permitted more detailed examination of signaling pathways expressed in the L-cell, although in some instances, information has been assumed by extrapolation from what is known regarding other neuroendocrine cells . An improved understanding of the intracellular mechanisms underlying GLP-1 secretion may lead to novel approaches to augment GLP-1 secretion in vivo, particularly if used in conjunction with agents designed to prevent the rapid degradation of GLP-1 in the circulation .
You May Like: Didn’t Get Third Stimulus Check
B Tissue Distribution Of The Glucagon Receptor
Both the sequences of glucagon and the glucagon receptor are highly conserved across mammalian species . Like GLP-1, binding of glucagon to the GCGR activates adenylyl cyclase through G proteins of the Gs subtype with subsequent generation of cAMP and activation of PKA and other downstream mediators as the major mode of intracellular signaling . Evidence for a GCGR was first demonstrated using hepatic membrane preparations , and the richest source of glucagon binding is in the liver and kidney . Lesser binding occurs in heart, adipose tissue, the CNS, adrenal gland, and spleen. GCGR gene expression is greatest in liver, with more modest mRNA levels in the kidney, heart, spleen, ovary, and the pancreatic islets . Consistent with the relative receptor expression, the liver and kidney play the major role in glucagon clearance, accounting for 70% of the removal from the circulation . Much of glucagon removal is through receptor-mediated endocytosis, but there is evidence for endovascular proteolysis by dipeptididyl peptidase IV and enzymatic degradation at the level of the plasma membrane . The half-life of glucagon in circulating plasma is relatively short: 2, 5, and 7 min in rats, dogs, and humans, respectively .
Medications Which Affect Glucagon Secretion
A number of medications have been developed to help modify the release of glucagon in type 2 diabetes.
Two different classes of diabetes drugs , DPP-4 inhibitors and incretin mimetics, act in response to the presence of meals to stimulate the increase of insulin and to inhibit the release of glucagon.
Read Also: Can I Still Get My First Stimulus Check
Measurements Of I By Digital Imaging Fluorometry
The superfusion chamber was placed on the stage of an inverted Nikon Diaphot microscope equipped with an epifluorescence illuminator and a ×40 oil immersion fluorescence objective . The chamber holder and the objective were maintained at 37°C. A 150-W xenon arc lamp and an Optoscan monochromator provided excitation light at 340 and 380 nm and emission was measured at > 515 nm by an intensified CCD camera . The Metafluor software controlled the monochromator acquiring fluorescence images of 30 accumulated frames at 340 and 380 nm every 4 s. i images were calculated from 340:380 nm ratio images as previously described .
Glucagon And Blood Glucose Control
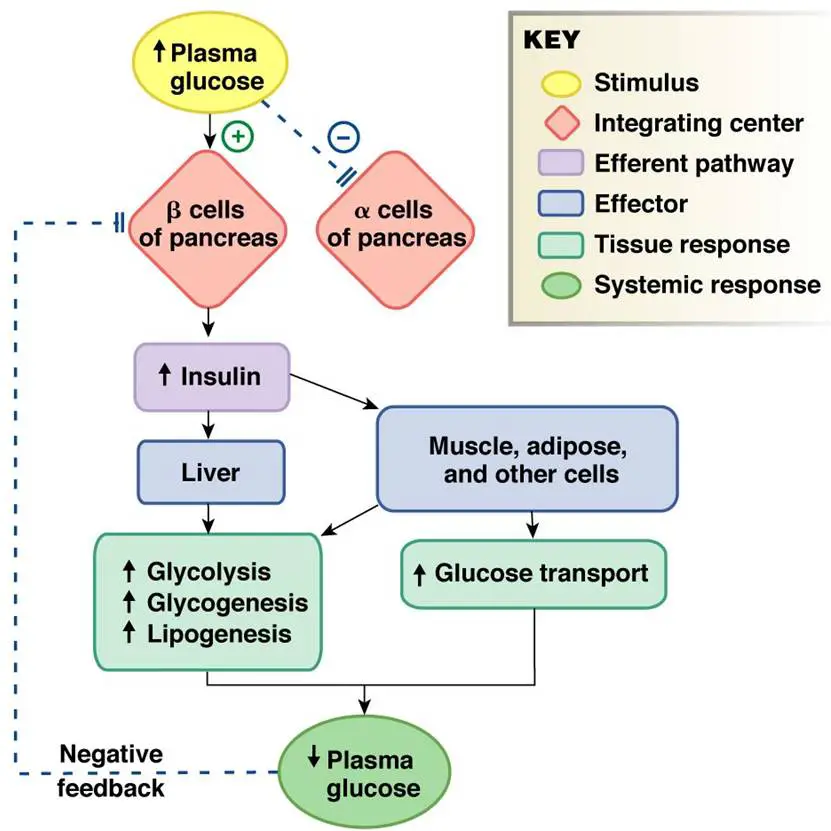
Glucagon is secreted by the alpha cells of the pancreatic islets in much the same manner as insulinexcept in the opposite direction. If blood glucose is high, then no glucagon is secreted.
When blood glucose goes low, however , more and more glucagon is secreted.
Like insulin, glucagon has an effect on many cells of the body, but most notably the liver.
The effect of glucagon is to make the liver release the glucose it has stored in its cells into the bloodstream, with the net effect of increasing blood glucose. Glucagon also induces the liver to make glucose out of building blocks obtained from other nutrients found in the body, such as protein.
You May Like: Stimulus Check Direct Deposit Date
Early Loss Of The Glucagon Response To Hypoglycemia In Adolescents With Type 1 Diabetes
Diabetes Care
Aris Siafarikas, Robert J. Johnston, Max K. Bulsara, Peter OLeary, Timothy W. Jones, Elizabeth A. Davis Early Loss of the Glucagon Response to Hypoglycemia in Adolescents With Type 1 Diabetes. Diabetes Care 1 August 2012 35 : 17571762.
To assess the glucagon response to hypoglycemia and identify influencing factors in patients with type 1 diabetes compared with nondiabetic control subjects.
Hyperinsulinemic hypoglycemic clamp studies were performed in all participants. The glucagon response to both hypoglycemia and arginine was measured, as well as epinephrine, cortisol, and growth hormone responses to hypoglycemia. Residual -cell function was assessed using fasting and stimulated C-peptide.
The glucagon response to hypoglycemia in adolescents with type 1 diabetes is influenced by the duration of diabetes and can be lost early in the course of the disease.
The purpose of this study was to 1) assess the glucagon response to both hypoglycemia and arginine as an independent stimulus in adolescents with type 1 diabetes with a range of diabetes duration, as well as in healthy control subjects, and 2) identify clinical and demographic factors that predict the glucagon response to hypoglycemia.
A Glucagon Antagonists And Agents That Reduce Glucagon Signaling
Blockade of glucagon action in humans has been possible with several small molecule GCGR antagonists developed by pharmaceutical companies, but the information available from these clinical programs is limited. One of these compounds, BAY27-9955, was demonstrated to block the actions of exogenous glucagon in nondiabetic subjects , but was not further developed and no information on treatment of diabetic subjects with fasting hyperglycemia or endogenous hyperglucagonemia was published. A small molecule, MK0893, lowered fasting glucose significantly in diabetic subjects in a dose-responsive manner, but these results were only presented in preliminary form . Similar findings have been reported with LY2409021, with dose-dependent reductions in fasting and postprandial glucose . As the pharmacological approach most analogous to genetic models of reduced GCGR signaling, antagonists have obvious potential as drugs to lower glucose and treat diabetes. However, the data from preclinical studies raise concerns that adverse effects such as -cell hyperplasia, hepatic steatosis, and susceptibility to hepatic toxicity could occur in treated patients. Moreover, the effects of these agents on glucose counterregulation would need to be carefully assessed.
Don’t Miss: How To Get Government Stimulus Check
Is There Anything Else I Need To Know About A Glucagon Blood Test
A glucagon blood test and a test called a glucagon stimulation test are both often called glucagon tests. But they are not the same thing. A glucagon blood test is used to check conditions related to glucose levels. A glucagon stimulation test checks for problems with growth hormones. It is most often used for infants and children.
A Pathophysiology Of Glucagon In Diabetes
Similar to insulin secretion, glucagon release in persons with diabetes is abnormal, and a number of defects have been identified from studies of diabetic subjects. In states of poorly controlled diabetes with severe insulin deficiency or ketoacidosis, plasma glucagon concentrations are extremely high . However, fasting hyperglucagonemia of a more modest degree is present in many T2DM patients even without metabolic decompensation, and a good case can be made that inappropriate fasting glucagon drives inappropriate HGP in diabetic patients . Similar to the diabetic -cell, the -cell in diabetic subjects has abnormal sensitivity to glucose with less suppression during hyperglycemia from enteral or parenteral sources, and plasma glucagon levels after eating mixed nutrient meals are generally higher with T2DM . In contrast, there is evidence for sluggish or reduced glucagon responses to hypoglycemia . It is not clear how this inappropriate response to low glucose is mediated, but the functional result suggests another form of -cell glucose insensitivity. There is emerging evidence that -cell dysfunction in diabetes has a genetic basis as a common polymorphism in the KIR6.2 gene, which predisposes to T2DM and is related to blunted glucose-induced suppression of glucagon .
Read Also: What About 4th Stimulus Check
Differential Dependence Of Exocytosis On Ca2+ Influx Through
The pancreatic A-cells are equipped with both -conotoxin-GVIAsensitive and nifedipine-blockable Ca2+ channels tentatively characterized as N- and L-type Ca2+ channels based on these pharmacological properties. We next explored the relative contribution of the different modalities of Ca2+ influx to depolarization-evoked exocytosis. When applied under control conditions , -conotoxin-GVIA reduced the integrated Ca2+ current by 18 ± 7% and exocytosis by 62 ± 5% . When applied at a concentration of 5 M , nifedipine reduced the integrated Ca2+ current to the same extent and yet only inhibited exocytosis by 11 ± 3% . Finally, as shown in Fig. 8 C, a 10-fold higher concentration of nifedipine , which can be expected to abolish L-type Ca2+-channel activity, reduced the whole-cell Ca2+ current by 68 ± 8% and exocytosis by 30 ± 7% .
Vi Regulation Of Insulin Release
Insulin release from pancreatic b cells is tightly regulated, and allows the sensitive response of insulin levels to calorigenic nutrients in the body. Glucose, free fatty acids, and amino acids serve as fuel stimuli for insulin release, promoting insulin granule exocytosis. Additional hormonal factors influence the regulation pathway. Pharmacological agents can also be used to augment insulin release.
A. Glucose-stimulated insulin secretion
Figure 5. Hyperglycemic clamp illustration. Example of hyperglycemic clamp testing in obese adolescents with normal glucose tolerance , impaired glucose tolerance , and type 2 diabetes . In the hyperglycemic clamp in healthy, non-diabetic individuals, glucose concentration is briskly elevated by administering a suitable intravenous glucose infusion at time 0. This elicits a rapid and short-lived insulin secretion peak due to release of preformed insulin vesicles, followed by a drop towards basal levels and then by a relatively rapid return to a sustained increase in insulin in the second half of the clamp as dextrose infusion is continued. This example illustrates the loss, in first and second phase insulin secretion, as individual progress from normal to impaired glucose tolerance, to type 2 diabetes. In the latter, the first phase insulin response is essentially lost and the second phase insulin response is reduced. ).
B. Proteins and Amino Acids
C. Lipids and Free Fatty Acids
D. Incretin Hormones
Read Also: Where Do I Put Stimulus Money On Tax Return